HTML
Review Article - (2021) Volume 11, Issue 8
An Innovative Approaches in Discovery of New Ways for Patient Targeting audit
Rishita Patel**Correspondence: Rishita Patel, Department of Pharmacology, Charutar Vidyamandal University, Gujarat, India, Email:
Received: 28-Sep-2021 Published: 19-Oct-2021
Abstract
The pharmaceutical industry and health professionals are experiencing a deficit in an innovation, especially in the finding new ways of patient targeting. Innovative advances in drug targeting discovery and includes Ethno pharmacology, biological function and holistic targeting provides an integral review and analysis of health, pathogenesis of disease and medicine effect and examine possible reasons behind the present catastrophe in drug discovery. The benefits of biological response and pharmacogenomics outcome and proponent the extension from disease centric discovery to patient-centric therapeutics involving chronobiotics, multi-target, whole systems approach. It also lays the path revive pharmaceutical revolution through a disciplined resurgence of antique medicine, appealing open innovation models and association trusted public-private partnerships. With impressive advances made in the evolution of biometrically relevant implements and technologies, the demand is higher and right now for a recommence commitment towards patients’ satisfaction. By absorbing real-life samples and state of the art reviews and cost effectiveness of product, this novel targeting methods provides profitable acumen into the learning and advancement strategies for healthcare professionals, academicians and learners in the pharmacy sciences and clinical practice.
Keywords: Drug discovery, Patient targeting discovery, Pharmacogenomics, Innovative approach, Network pharmacology, Holistic lifestyle, Chronotherapeutic.
Abbreviations: NP: Network Pharmacology; ABP: Ambulatory Blood Pressure; FDA: Food and Drug Administration; IBD: Inflammatory Bowel Syndrome; CD: Crohn’s Disease; RA: Rheumatoid Disease; SCD: Sickle Cell Disease; RBC: Red Blood Cell; NAD: Nicotinamide Adenine; Dinucleotide NADH: Nicotinamide Adenine Dinucleotide Hydrogen; RGP: Rigid Gas Permeable; Ortho-K: Orthokeratology; CRT: Corneal Refractive Therapy; GVSS: Gentle Vision Shaping System GVSS; ADFs: Abuse-Deterrent Formulations; PFDDM: Patient Focused Drug Development Meeting; NP: Network Pharmacology; KEGG: Kyoto Encyclopedia of Genes and Genomes; HDN: Human Disease Network; MDN: Metabolic; Disease Networks; PDN: Phenotypic Disease Networks; HSDN: Human Symptom Disease Network; DTN: Drug Target Network; API: Active Pharmaceutical Ingredient.
Introduction
Prescription scientific and technological evolution over the last 25 years, especially in omics (studying of biology), high performance screening technologies, translational/combinational chemistry, instrumentation and medical imaging have opened exciting paths for the discovery of new drug candidates. Even so, major difficulties remain and are constantly evolving. The extent of new research is falling and numerous drugs approved by regulators are being withdrawn due to toxicity and safety issues. The drug discovery process is facing severe bottleneck and innovation deficit, which is putting the pharmaceutical industry under severe pressure. Chemical, biology and genomic outlook, primarily monogenic, based on one target-one gene-one disease is not showing optimum results [1]. Therefore, there is a requirement for innovative ways for patient targeting discovery based on ethno pharmacology, holistic targeting and human biology system.
Single-target single-drug paradigms have long dominated the drug discovery and development landscape. While advantageous drug relevance towards a target is useful against diseases that are heritable, the administration of single-drugs has been less rewarding against the widely prevalent diseases affecting huge population groups like cancer, cardiovascular diseases, metabolic insufficiency and neurological problems. Maximum of these complex diseases are multifactorial and are directed by genetics, environment, age and sex. Although drug combinations directed against two or more targets are widely employed in treatment of some diseases, the polypharmacology of new molecular entities is the new emerging prototype for the targeting jumbled complex diseases [2].
Most of the drugs recalled or withdrawn over the last half a century are associated with toxic effects on different organs and systems. Among various effects leading to drug recall or withdrawal, hepatic toxicity ranks the highest followed by cardio toxicity. Specificity to a particular target may not ensure that a drug has no effect on other targets. A metabolic network study of thalidomide, statins, and troglitazone drugs shows the involvement of on-target pharmacological and off-target nonspecific unwanted effects. Drug toxicity is due to cumulative effect of target modulation and intrinsic property of drugs, known as “toxicity triangle effect”.
The one-drug/one-target/one-disease approach to drug discovery is presently overlook many exceptions of safety, efficacy and defendable. Network biology and poly-pharmacology approaches got valuing recently as methods for omics data integration and multitarget drug development and effectiveness respectively. Network Pharmacology (NP) that works at the effect of drugs on both the interactive and the human disease network level. Ayurveda, the ancient system of Indian medicine, uses intelligent formulations containing wide ingredients and different bioactive compounds; however, the scientific standards and mechanisms remain largely undiscovered. NP approaches can serve as a valuable standard for evidence-based Ayurveda to understand the medicines’ supposed actions, direction of use and mechanisms. NP and its potential to discover ancient medicine systems to overcome the drug discovery imposes is important to explore [3].
Vaccines have provided some of the greatest successes in the history of medicine, including the eradication of smallpox and we’re now reaching to the eradication of polio. However, as same as polio, successes have not been achieved with vaccines against global killers like, Corona, AIDS, tuberculosis, malaria, and different infectious diseases. This has resulted in an important shift in research approaches and an increase in the role of systems finding. Advanced technologies and holistic targeting of immune responses are being followed for prophylactic and therapeutic vaccines against infectious diseases, cancer, and autoimmune indications. Systems approaches are enabling newer roles for adjuvants and delivery systems in optimum targeting of protective immune responses. Immunomodulatory agents derived from botanicals are under consideration as novel adjuvants. The scope and finding offered by chemical diversity of botanical immune drugs towards research of new vaccine adjuvants are highly entrancing [4].
Typically, a drug is any chemical moiety that prevents, alleviates, cures, and manages disease with the fundamental goal of regaining health. Deception of pharmaceutical drugs in clinical practice overlooks concepts of homeostasis and interplay of biological networks. Holistic management of the patient involves dietary modifications, exercise, counseling, therapeutic procedures and behavioral modifications. Ongoing and emerging research on lifestyle and behavioral interventions suggests several mechanisms those can reduce and replace use of medicines. Ayurveda and yoga suggest holistic management with triad of drugs, diet, and lifestyle. Lifestyle and behavior management holds importance as therapeutic interventions but are more beneficial for risk reduction, disease prevention, and health promotion. Lifestyle research can give safer, affordable, on hand and optimistic paradigm for management and even reversal of non-communicable diseases. The latest research in psycho-neuro immune-endocrinology shows meditation can act like drugs and has the potential to modulate several physiological functions. The shift from medication to meditation will be the future of medical science [5].
Objectives
1. Scrutinize the reasons of historical drug failures to provide valuable recognition and understanding of lessons.
2. Application of current scientific output to enhance learning from ancient knowledge systems and through the interconnection of traditional and novel medicines.
3. Discusses progress in technologies and system biology to upright the transition from formulation discovery to therapeutic discovery [6].
Literature Review
New ways for patient targeting are enlisted below
1) Holistic drug targeting approach-curcumin, the holistic Avant-grad, ethno pharmacology
2) Chronotherapeutic
3) Information Collection through mobile device application-use for effective and safe drug targeting
4) New formulation-not novel but has important medical value as per patient’s need
5) Approaching development of new therapies
• Biomarkers
• Biosimilars
• Rare Disease Treatment-sickle cell disease
• Self-Injected Devices • Developing new treatment that can ultimately provide a cure
6) Patient focused drug development meeting
7) Network Pharmacology (NP)-multi target drug development
8) Vaccines and immune drugs discovery-prophylactic and therapeutic vaccines
9) Holistic lifestyle
Holistic drug targeting approach-curcumin, the holistic avant-grad, ethnopharmacology
1. Curcumin is a polyphenol extract from rhizomes of turmeric has been used in Ayurveda and ancient traditional medicine from ancient times. It is highly pleiotropic molecule; Safety and efficacy of curcumin has been demonstrated in cell-based studies and in animal models and humans for a wide range of diseases [7-10].
2. Curcumin used either as a single agent or in combination with other agents.
3. Modern science has described the molecular form of the pleiotropic activities of curcumin. Anticipating curcumin’s multi-targeting nature and its effectiveness against multiple human diseases, polyphenol the active molecular structure of curcumin appears to represent a holistic medicine pointing to the entire human body.
4. The pleiotropic activities of curcumin acquired from its ability to modulate signaling pathways either by direct binding to molecules or in an indirect manner and that coded gene expresses in number of cells at a same time showing its multi-targeting activity [11,12].
5. Its nature is multi-targeting and efficacious in multiple human diseases like cancer, metabolic syndromes, infections, healing properties.
6. Increased use of traditional medicinal products and Wide spread demand have generated public health challenges globally in terms of quality and safety. There has been remarkable advancement in science and technology in the past few decades, and to serve to rapidly changing global health demands due to globalization and changing lifestyle, these technologies can provide newer, costeffective, and supportive ways to judge and monitor quality and safety of the products. The potential utility of extending modern approaches of safety monitoring for botanicals as shown in Figure 1 have been reviewed [13,14].
Ethnopharmacology
1. Ethnomedicine is a study or comparison of the traditional medicine based on bioactive compounds in plants and animals and practiced by various inherited groups, especially those with little access to western medicines, e.g., ancient peoples [15].
2. Ethnopharmacology is a related study of racial groups and their use of plant compounds. As traditional medicine is a source of lead compounds for drug discovery, it is linked to medicinal plant use and ethno botany. Importance has long been on traditional medicines, although the approach has also proven usefulness to the study of modern pharmaceuticals [16,17].
3. It involves studies of the:
• Identification and ethnotaxonomy (cognitive categorization) of the natural material, from which the candidate compound will be produced.
• Traditional preparation of the pharmaceutical forms.
• Bio-evaluation of the possible pharmacological action of such preparations (ethnopharmacology).
• Their potential for clinical effectiveness.
• Socio-medical aspects implied in the uses of these compounds (medical anthropology).
The opium poppy from Papaver somniferum, used in traditional medicine for millennia, is the source of the alkaloids and codeine as shown in Figure 2.
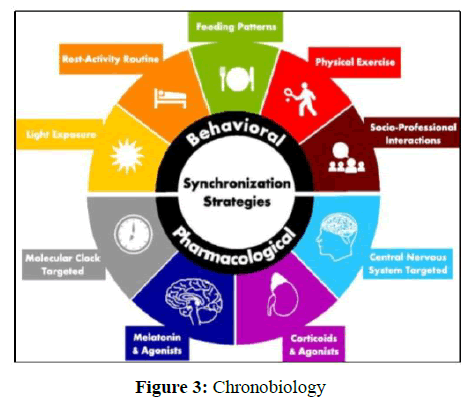
Chronotherapeutics
1. Chronotherapeutic refers to manage a patient according to a person's daily, monthly, seasonal, or yearly biological clock, in order to maximize the health benefits and minimize side effects of drugs [18-20].
2. It is novel and innovative concept for discovery of new ways of patient targeting. it is related to biological rhythmicity of an individual. By finding chrono biotics there will be new hope for patient targeting drug delivery system (Figure 3).
3. Biological Rhythms can be of four types.
• Circadian rhythm: it lasts only for 24 hrs.
• Ultradian rhythm: it is spontaneous in nature but have very short duration.
• Diurnal rhythm: it depends on day and night cycle
• Infradian rhythm: it lasts more than 24 hr.
4. Bedtime BP treatment improves CVS risk reduction. The Hygia Chronotherapy trial report was published in 1st
5. October 2019 by FDA.
6. Conclusion of trial shows routine ingestion by hypertensive patients of ≥ 1 prescribed BP lowering medications at bedtime, as opposed to upon waking, results in improved ABP control (significantly enhanced decrease in asleep BP and increased sleep-time relative BP decline, i.e., BP dipping) and most importantly, markedly diminished occurrence of major CVD events.
7. Larger number of disease symptoms depends on circadian rhythm of human body. This is the interesting area where researcher can focus and find out ways for future targeting therapies. This can be the best way for future concept of personalized medicine or personal medicine [21-24].
Information collection through mobile device application-use for effective and safe drug targeting
1. Now a day’s various mobile applications are available which make comforts for patients to remember the medicines. Various nutrition apps, health apps, cardio apps are available to be get healthy.
2. Some apps like pill reminder app which make elderly patients to remember their daily medications. Medication tracker app, patient portals, healthify me app and period tracking app etc are available which make patients to adhere with their therapy [25,26].
3. Period tracking app helps women to track periods for all months or changes in that also incorporated in the apps, so it is really good application for getting information of menstrual periods and making pregnancy plan.
4. Figure 4 represents that it is best and informative device to manage safe and effective targeting various patient problems.
New formulation-not novel but has important medical value as per patient’s need
1. Formulated drugs are stored in container closure systems for extended periods of time. These include blisters, bottles, vials, ampules, syringes, and cartridges. The containers made from a variety of materials such as glass, plastic, and metal and the drug may be stored as a solid, liquid, or gas. That’s why it is important to check whether there are any undesired interactions between the preparation and the container
2. Here I am coding one example of new formulation of allergic response (hypersensitivity) is Symjepi which is new device marketed by Adamis pharmaceuticals approved by FDA in 2019.
3. Epipen was approved by FDA for the allergic reaction marketed by Mylan. Both use for type-I allergic hypersensitivity reaction. Use by intramuscular/subcutaneous route.
4. Epipen VS Sympeji contains epinephrine use in asthma, immunology.
5. Epipen (Mylan) is lager and multidose containing device and has high cost while Symjepi (Adamis) has lower cost, smaller size and single dose unit with user friendly device.
Approaching development of new therapies
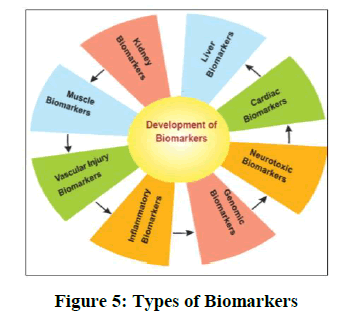
Biomarkers: Biomarkers is an object that evaluated as an indicator of normal biological processes, pathogenic processes and pharmacological response of therapeutic use of drug.
1. They are a measurable indicator of the severity and presence of some disease as shown in Figure 5.
2. Biomarkers use for
• Disease monitoring
• Disease prognostication
• For treatment response
• For monitoring toxicity
3. It improves the efficiency of product development and helps identify safety problems. Biomarkers available for cancer, blood, vascular disease and heart disease.
4. FDA approved biomarkers
• Safe-t consortium for drug induced vascular injuries. It is safety biomarker to detect risk of vascular injuries.
• Fossa consulting developed biomarker beat to beat restitution as assessed by ECG. It is safety biomarker for arrhythmia risk [27,28].
Biosimilars: A bio-similar is a biologic medical product (also known as biologic) highly similar to another approved biological medicine (the 'reference medicine').
1. Biosimilars are approved in consonance with the same standards of pharmaceutical quality, safety and efficacy that apply to all biological medicines.
2. Biosimilars are officially approved form of original "innovator" products and manufactured when the original product's patent expires. Reference to the innovator product is an integral component of the approval.
3. Despite that heterogeneity (high molecular complexity, highly sensitive for changes in manufacturing processes) all biopharmaceuticals, including biosimilars, must maintain consistent quality and clinical performance throughout their lifecycle.
4. A bio-similar is not considered as a generic of a biological medicine. Mostly because of the natural variability and more complex manufacturing of biological medicines do not allow an exact replication of the molecular microheterogeneity.
5. Drug-related authorities such US's Food and Drug Administration (FDA), and the Health Products and Food Branch of Health Canada hold their own guidance on requirements for demonstration of the similar nature of two biological products in terms of safety and efficacy.
6. According to them, analytical studies demonstrate that the biological product is highly similar to the reference product, despite minor differences in clinically inactive components in the animal studies including the assessment of toxicity, and a clinical study or studies including the assessment of immunogenicity and pharmacokinetics or pharmacodynamics.
7. These products are sufficient to demonstrate safety, purity, and potency in one or more appropriate conditions of use for which the reference product is licensed and is intended to be used and for which licensure is sought for the biological product. They are biologic medical product highly similar with approved medicine.
8. Biological medication is able to target the cause of disease in ways that were not previously possible allowing patient lives healthier and more active lives.
9. Biosimilars are available for: IBD, CD, RA, Asthma, Multiple sclerosis.
• Brand name of biologics is reference product
• Highly similar biologics known as bio-similar
10. Avsola bio-similar for Remicade reference drug contain Infliximab for the treatment of IBD AND CD by USFDA in December 2019 [29].
Rare disease treatment-sickle cell disease
Sickle cell disease: CDER approved treatment for 20 years back disease of SCA ENDARI (L-GLUTAMINE ORAL POWDER) in 2017
1. As per Figure 6 Endari (L-glutamine oral powder) reduces oxidant damage to red blood cells by improving the redox potential of nicotinamide adenine dinucleotide (NAD), a coenzyme that has been identified as the primary regulator of oxidation.
2. It is a non-essential amino acid present profusely throughout the body and is involved in many metabolic processes. L-glutamine is synthesized from glutamic acid and ammonia. L-glutamine is the principal carrier of nitrogen in the body and is an important energy source for number of cells.
3. An oral formulation of L-glutamine was approved by the FDA in July 2017 for use in sickle cell disease. This oral formulation is marketed under the tradename Endari by Emmaus Medical [30,31].
4. How the amino acid L-glutamine is treated sickle cell disease (SCD) is not fully understood. Oxidative stress is an important factor that is involved in the pathophysiology of SCD. Sickle red blood cells (RBCs) are more susceptible to oxidative damage than normal RBCs, which may result in the chronic hemolysis and vaso-occlusive events associated with SCD. The pyridine nucleotides, NAD+ and its reduced form NADH, play important roles in regulating and preventing oxidative damage in RBCs.
5. L-glutamine may improve the NAD redox potential in sickle RBCs through increasing the availability of reduced glutathione [32].
Hemophilia-A: A medical condition in which the ability of the blood to clot is severely reduced, causing the sufferer to bleed severely from even a slight injury. This serious condition is typically caused by a hereditary lack of a coagulation factor, mostly lacking of factor Ⅷ
1. FDA approved non blood product HEMLIBRA shown in Figure 7 prevent bleeding by developing antibodies called factor Ⅷ inhibitors in 2017.
2. Hemlibra® contains bispecific factor Ⅻ a and factor Ⅹ directed antibody. These restore the function of missing activated factor Ⅷ, which is necessary for effective haemostasis of clotting. The drug is available in 150 mg/ml dose for subcutaneous use (Figure 7).
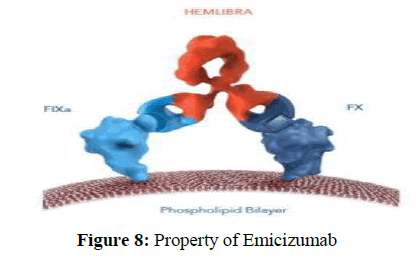
3. Emicizumab exerts its action by performing the function of the coagulation Factor Ⅷ without presenting a structural homology. It presents a dual specificity which allows it to bind to both the Factor Ⅸ and Factor Ⅹ, performing the required bridging activity for the launch of the coagulation cascade [33].
4. As per Figure 8 Emicizumab is a humanized recombinant monoclonal antibody that increase the function of the coagulation Factor Ⅷ and it has the capacity to bind simultaneously to activated Factor Ⅸ and Factor Ⅹ. The ability of Emicizumab to bind to all these three different factors allows it to overcome immunogenicity and unstable hemostatic efficacy produced by previous Factor Ⅶ agents. Emicizumab was originated as an improved form of hBS23 and it was approved on November 16, 2017. It was created by Chugai Pharmaceuticals Co. Ltd.and co-developed with Roche and Genentech.
Self-injected devices
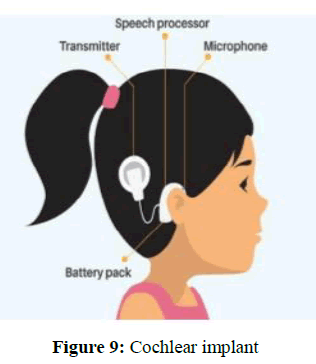
Cochlear implants: A cochlear implant is a small, complex electronic device that can help to provide a sense of sound to a person who is profoundly deaf or severely hard-of-hearing. The implant made up of an external portion that fits behind the ear and a second portion that is surgically placed under the skin as per shown in Figure 9.
As per Figure 9 the implant has two main components. The outside component is generally worn behind the ear, but could also be attached to clothing, for example, in young children. This component is also known as the sound processor that contains microphones, electronics that include digital signal processor chips, battery, and a coil which transmits a signal to the implant across the skin. The inside component which is actual implant, has a coil to receive signals, electronics, and an array of electrodes which is placed into the cochlea, which stimulate the cochlear nerve. The surgical procedure is performed under general anesthesia. Surgical risks are minimal but can produce buzzing of ear, facial nerve bruising and dizziness [34].
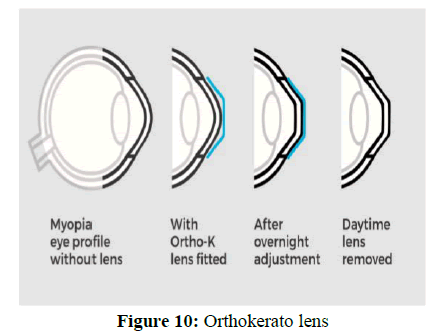
Contact lenses: New device which approved by FDA is orthokeratology with RGP (rigid gas permeable) lenses which can wear during night. Lenses reshape the front surface of eye. It reshapes cornea in night and vision is clear after removing lenses during whole day.
1. Orthokeratology (also referred to as Ortho-K, OK, overnight vision correction, Corneal Refractive Therapy (CRT) and Gentle Vision Shaping System (GVSS) refers to the use of gas-permeable contact lenses that for short term reshape the cornea to reduce refractive errors such as myopia, hyperopia and astigmatism.
2. Orthokeratology can be used as optional to eyeglasses, refractive surgery, and for those who prefer not to wear contact lenses during the day. Orthokeratology is often used for people with up to -6.00 Diopters of myopia; though exceptions may be made if the prescription is only slightly lower (e.g., -6.25).
3. Tissue growth and proliferation studies indicate that epithelial cells adjust growth in response to presence of a foreign material. CRT lens stimulate cell growth where less proximity coincides with added growth and more proximity coincides with growth repression there by creating a 'proximal pressure' on the epithelial cells.30,31
4. The mechanism behind ortho-k is that around 60% of the eye's focusing power is provided by the cornea, and this is extremely sensitive to very small changes: 6 μ m flattening of corneal thickness (around 5% of the thickness of a human hair) results in 1 Diopter of changed vision in myopia.
5. Hence, a specially shaped lens can be used to lightly press the cornea, causing it gradually to be reshaped to the correct shape for focused vision. The corrective effect lasts up to 72 hours once initially acclimatized, which is long enough to be a practical means of eyesight correction. Ortho-K reduces future or ongoing eyesight changes or increased myopia, although it is being researched (Figure 10).
6. These lenses are safer than ordinary contact lenses, since they are typically worn for much shorter periods (6-8 hours rather than daytime or 24/7) and while asleep rather than while active, so risk with these lenses is very less. Surgical process is not required, corrections to the eye's shape after surgery can be handled over time because surgery corrects vision at a single point in time, but postoperative ongoing changes to eyesight will continue to occur during the patient's lifetime. It is generally safe for younger patients. Ortho-K contact lens safety generally as same as with all contact lenses, one is required to maintain good cleaning and hygiene discipline. Rigid lenses are considered to be not as comfortable as soft lenses, but Ortho-K is very soft in nature therefore it avoids the feeling during waking hours of the eyelids moving over the lens edges while blinking [35,36].
Developing new treatment
1. Developing new treatment is new alternate approach to improve patient targeting.
2. Abuse deterrent formulation.
3. For severe intractable pain, opioids always remain a critical and appropriate component of treatment. However, abuse, misuse, and diversion of prescription opioids are significant public health concerns. For that opioid Abuse- Deterrent Formulations (ADFs) are made for opioid risk management plan to manage patient’s pain relief and quality of life while giving protection against potentially harmful consequences of opioids from misuse and abuse. Opioid ADFs are constructed to make manipulation more difficult and administration via non-oral routes less appealing. Under US Food and Drug Administrationapproved ADF labeling, there are currently nine extendedrelease and one immediate-release opioid pain medications.
4. Most of all uses physical barriers or agonist/antagonist combinations to deter manipulation and abuse. Evidence suggests that opioid ADFs decrease rates of abuse and diversion of opioids in the USA; however, some opioid ADFs are not yet commercially available long enough to undergo post-marketing data analyses. Opioid ADFs along with the use of prescription drug monitoring programs, clinical assessment tools, toxicology testing, and coprescribing of naloxone are all tools that can be used to reduce opioid abuse.
5. Patient education on the risks of abuse and diversion is much more important and includes a discussion of appropriate use of medication and proper storage.
6. Physician and nurse are plays key role in battling opioid abuse and play a key role in recognizing and mitigating the risks of prescription opioid diversion, abuse, and misuse (Intentional and unintentional) and in identifying patients at risk for abuse while still providing pain relief to patients.
It will decrease product abuse potential and still being able to provide safer and accurate delivery of the opioids for therapeutic benefit as per shown in Figure 11.
7. EMBEDA (morphine sulfate and naltrexone hydrochloride) extended-release capsules are very good ADF formulation which is use for the management of pain severe enough to require daily opioid , around-the-clock and long-term treatment purpose and for which alternative treatment options are inadequate [37,38].
Patient focused drug development meeting
1. FDA took initiative to hear from patient from perspective of their condition and available treatment option. Voice of the patient recorded, written comment and unique views taken which is used for patient focused drug development meeting.
2. For alopecia areata pfddm were done in 2017 by FDA which is predominant on skin and affects nails also.
3. Medication used is minoxidil, local steroids, immunotherapies (Figure 12).
Network Pharmacology (NP)-multi target drug development
1. Network science application towards identifying, preventing, and treating diseases. This branch focuses on using network topology and network dynamics in identifying diseases and developing medical drugs. Biological networks, like protein-protein interactions and metabolic pathways, are core focus for network medicine. Disease networks plays crucial role which map relationships between diseases and biological factors. Epidemiology is extensively studied using NP. Medically focused area of systems biology is a NP.
2. The whole set of molecular interactions in the human cell (interactome), used for disease diagnosis and prevention. Protein-protein interactions have been mapped, using proteins as nodes and their interactions between each other as links. These maps utilize as a database such as Bio GRID and the human protein reference database. The metabolic network encloses the biochemical reactions in metabolic pathways, connecting two metabolites if they are in the same pathway [39].
3. Diseases can be classified not by their principle phenotypes (pathophenotype) but by their disease module, which is a neighborhood or group of components in the interactome that, if disrupted, results in a specific pathophenotype. Disease modules can be used in a variety of ways, such as predicting disease genes that have not been discovered yet. Therefore, network pharmacology looks to identify the disease module for a specific pathophenotype using clustering algorithms. Using interactome networks, scientist can discover and classify diseases, as well as develop treatments through knowledge of its associations and their role in the networks.
4. The diseasome are networks in which the nodes are diseases and the links, the strength of correlation between them. This connection is frequently measured based on associated cellular components that two diseases share. The first-published human disease network (HDN) looked at genes, finding that many of the disease associated genes are non-essential genes, as these are the genes that do not completely disrupt the network and are able to be passed down generations.Metabolic disease networks (MDN), in which two diseases are connected by a shared metabolite (metabolic pathway) have also been comprehensively studied and is more specifically relevant in the case of metabolic disorders.
1. Three representations of the diseasome are:
• Shared gene hypocrisy tells that if a gene is linked to two different disease phenotypes, then the two diseases likely have a common genetic origin (genetic disorders).
• Shared metabolic pathway hypocrisy tells if a metabolic pathway is linked to two different diseases, then the two diseases likely have a shared metabolic origin (metabolic disorders).
• Disease comorbidity formalism uses phenotypic disease networks (PDN), where two diseases are linked if the observed comorbidity between their phenotypes exceeds a predefined threshold. This does not look at the mechanism of action of diseases, but captures disease progression and how highly connected diseases correlate to higher mortality rates.
2. Etiome is networks of environmental and genetic etiological factors linked with shared diseases, can be also used to assess the clustering of environmental factors in these networks and understand the role of the environment on the interactome. The human symptom-disease network (HSDN), published in June 2014, showed that the symptoms of disease and disease associated cellular components were strongly correlated.
3. Network pharmacology is a emerging field based in pharmacology that looks at the effect of drugs on both the interactome and the diseasome. The drug-target network (DTN) plays an important role in understanding the mechanisms of action of approved and under examination drugs. The network theory view of pharmaceuticals is based on the effect of the drug in the interactome, especially in the region of drug target. Combination therapy for a complex disease (poly-pharmacology) is suggested in this field since one active ingredient (API) aimed at one target may not affect the entire disease module. The concept of disease modules can be used to aid in drug discovery, drug design, and the development of biomarkers for disease detection. There are a variety of ways to identifying drugs using network pharmacology; a simple example of this is the "guilt by association" method. This states if two diseases are treated by the same Drug, a drug that treats one disease may treat the other. Drug repurposing, drug-drug interactions and drug side effects have also been studied in this field.
Vaccines and immune drugs discoveryprophylactic and therapeutic vaccines
1. A therapeutic vaccine is a vaccine which is administered after the disease or infection has already occurred. The therapeutic vaccine works by activating immune system of the patient to fight towards different newly and existed infection. The difference in therapeutic vaccine and vaccine is that vaccines are administered to individuals as a precautionary measure to avoid the infection and disease while therapeutic vaccines are disease and infection. Therapeutic vaccine fights the existing infection in the body rather than immunizing the body for protection against future diseases and infections. The therapeutic vaccines are mostly against viral infections. The patients affected with chronic viral infections are administered with therapeutic vaccines, as their immune system is not able to produce enough efficient antibodies.
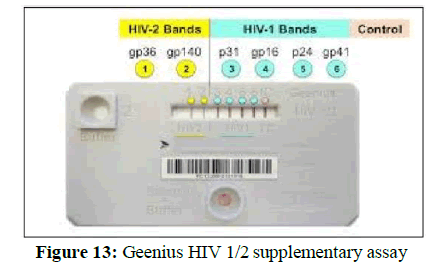
2. Provenge, developed by Dendreon, was the first therapeutic vaccine approved by FDA in 2010. This therapeutic vaccine helped in treating prostate cancer where patients own white blood cells (WBCs) were taken and treated with drug (vaccine) to train them to differentiate and fight cancer cells (Figure 13).
3. Figure 13 shows geenius HIV 1/2 supplementary assay for HIV Type-1 and 2. It’s uses for confirmation and the geenius HIV 1/2 supplemental assay is a single-use immunochromatographic assay for the confirmation and differentiation of individual antibodies to human immunodeficiency virus types 1 and 2 (HIV-1 and HIV-2) in serum, or plasma samples, lithium heparin, sodium citrate, and CPD) from blood donors.
4. The geenius HIV 1/2 supplemental assay is intended for use as an additional, more specific test for human
5. Serum and plasma samples with repeatedly reactive results by an FDA licensed blood donor screening test for antibodies to HIV‑1/HIV‑2. The results of the geenius HIV 1/2 supplemental assay are read and interpreted only with the geenius reader with dedicated software [40].
Holistic lifestyle
1. Living a holistic lifestyle is a lot healthier among every style of living.
2. Holistic lifestyle means choosing to live your life and heal your body through natural remedies. You observe everything as a whole is uniqueness about this way of living.
3. This all following are the points to follow to live happy and healthiest life.
• Practice mindfulness.
• Recognize and respect the powers of your body.
• Eat clean foods.
• Focus on positive relationships.
• Recycle energy.
• Let yourself grow.
• Love life, and be respectful.
• Connect.
• Be aware.
• Meditate.
• Be active.
Conclusion
This review shows such an area that needed to be discovered more and should be focused to create a new innovative approach for patient focused targeting. Working on this explored way will improve better patient targeting with improved specification. This will optimize response with minimization of toxic effect of drugs. If Patients are benefited as per their prime need only then discovery is known as innovative.
Acknowledgment
I am here thanking to almighty “God” and my guru “Mahant swami Maharaj” for in ti Thus it concludes that this innovative targeting method provides profitable awareness in learning advancement strategies for healthcare professionals, academicians, and learners in the field of pharmacy sciences and clinical practice me completion of my work.
Conflict of Interest
The author declares none.
References
- Patwardhan B, Chaguturu R. J. Ayu. Integ. Med. 2017; 10(2): 143-145.
- Acharya D, Shrivastava A. Aavishkar. Pub. 2008.
- Rochow VBM. J. Ethnobio. Ethnomed. 2017; 13 (9):53-57.
- Homas MJ, Carolyn FS. Praeger. Pub. 1996.
- Buer JK. J. Inflammopharmacol. 2014; 22 (5): 263-267.
- Chronotherapy. Encyclopedia. 2021.
- Ramon CH, Juan JC. J. Eur. Hrt. 2020; 41 (48): 4565-4576.
- Kumar A, Chen F, Mozhi A. J. Nanoscale. 2013; 5 (18): 8307-8325.
- Pharmaceuticals corporation. Adamis. 2020.
- Strimbu K, Jorge AT. J. Curr. Opin. HIV. AIDS. 2010; 5 (6): 463-466.
- Encyclopedia. Biomarkers. 2021.
- Anthony A F. J. Ann. Noninv. Electrocardiol. 2017; 22 (5).
- U.S. Food and drug administration. Biosimilar. 2021.
- U.S. Food and drug administration. Biosimilar. 2020.
- Skingle D. J. Rheu. Musculoskel. Dis. 2015
- Nick C. J. Pharm. Med. 2012; 26 (3): 145-152.
- Lamanna WC, Holzmann J, Cohen HP, J. Exp. Opi. Bio. Ther. 2018; 18 (4): 369-379.
- Amgen. Biosimilar. 2021.
- U.S. Food and drug administration. biosimilar. 2018.
- Oldenburg J, Johnny NM, Kim B. N. Engl. J. Med. 2017; 377(9): 809-818.
- Hanabusa M, Taki H, Matsushita M, Sato T, Fukutake T, Fukazawa K. Engl. J. Med. 2016; 374 (21): 2044-2053.
- U.S. Food and drug administration. Drug. 2018.
- Shimonishi N, Nogami K, Ogiwara K. J. Haem. 2020; 26 (3): 97-105.
- U.S. Food and drug administration. Drug. 2017.
- National institute on deafness and other communication disorders. Health. 2021.
- Chan SY. Loscalzo J. J. Cir. Res. 2012; 111 (3): 359-374
- Goh KI, Cusick ME, Valle D, Childs B, Vidal M, Barabasi AL. Nat. Acad. Sci. 2007; 104 (21): 8685-8690.
- Barabasi AL, Gulbahce N, Loscalzo J. J. Nat. Rev. Gen. 2011; 12 (1): 56-68.
- Loscalzo J, Barabasi AL, J. Sys. Bio. Med. 2011; 3 (6): 619-627.
- Rual JF, Venkatesan K, HaoT, Hirozane-Kishikawa T, Dricot A, Vidal M. J. Nature. 2005; 437 (7062): 1173-1178.
- Ravasz E, Somera AL, Mongru DA, Oltvai ZN, Barabasi AL. J. Sci. 2002; 297 (5586): 1551-1555
- Braun P, Rietman E, Vidal M. J. Nat. Acad. Sci. 2008; 105 (29): 9849-9850
- Hidalgo CA, Blumm N, Barabasi AL, Christakis NA. J. Plos. Comp. Bio. 2009; 5 (4): 56-79.
- Liu YI, Wise PH, Butte AJ. J. BMC. Bioinfo. 2009.
- Zhou X, Menche J, Barabasi AL, Sharma A. J. Nat. Comm. 2014.
- Hopkins AL. J. Nat. Chem. Bio.2008; 4 (11): 682-690.
- Patwardhan B, Chaguturu R. J. Ethnopharmacol. Sys. Bio. Holi. Targ. 2017.
- Rich R, Fleishe A, William T, Gillespie LS, Paul ME, Chinen J, Shearer WT. J. Elsevier. 2013; 465-479.
- James G. J. Hum. Vac. Immun. 2012; 9 (1): 219-221.
- U.S. Food and drug administration. Vaccines. Blood. Biologics. 2019.
Manuscript Submission
Submit your manuscript at Online Submission System
Google scholar citation report
Citations : 1101
International Journal of Pharmacy received 1101 citations as per google scholar report
International Journal of Pharmacy peer review process verified at publons
Indexed In
- CAS Source Index (CASSI)
- HINARI
- Index Copernicus
- Google Scholar
- The Global Impact Factor (GIF)
- Polish Scholarly Bibliography (PBN)
- Cosmos IF
- Open Academic Journals Index (OAJI)
- Directory of Research Journal Indexing (DRJI)
- EBSCO A-Z
- OCLC- WorldCat
- MIAR
- International committee of medical journals editors (ICMJE)
- Scientific Indexing Services (SIS)
- Scientific Journal Impact Factor (SJIF)
- Euro Pub
- Eurasian Scientific Journal Index
- Root indexing
- International Institute of Organized Research
- InfoBase Index
- International Innovative Journal Impact Factor
- J-Gate